 
The formal charge being a theoretical concept does not indicate any real charge segregation in the molecule. The relationship between the amount of bonding electrons and how many are technically “owned” by the atom is explicitly stated in this idea. When more than one plausible Lewis’ structure exists, the idea of formal charges can be utilized to determine the most acceptable Lewis’ structure. In exceptional cases, more than one valid Lewis structure for molecules and polyatomic ions is discovered. Significance of Calculating Formal Charge The formal charge is only a helpful bookkeeping procedure it does not indicate actual charges. The calculated formal charge is not the actual charge of the atom. Furthermore, the sum of the formal charges in an ion should equal the charge of the ion. 
The sum of formal charges of all atoms in a molecule must equal to zero. Because bonding e– is shared by two atoms, a multiplier of 1/2 is applied to the number of bonding e– when calculating formal charge analytically. 
It is a theoretical charge over a single atom of an ion since the real charge over a polyatomic molecule or ion is dispersed throughout the entire ion rather than a single atom. It is the difference between the valence electron of that atom in the elemental state and the number of electrons assigned to that atom in the Lewis structure. If put in another way, when we take the number of valence electrons of a neutral atom, subtract the nonbonding electrons, and subtract the number of bonds connected to that atom in the Lewis structure, a formal charge on the atom is obtained. The formal charge definition can be elaborated as the hypothetical charge on the atom if the electrons in the bonds are distributed evenly between the atoms. N is the number of non-bonding valence electrons on this atomī is the total number of electrons participating in bonding with other atoms in the molecule. V the number of valence electrons in the ground state of the atom The following equation can calculate the formal charge of an atom in a molecule: The formal charge can be calculated by excluding the number of electrons in the lone pairs and the number of bonds from the total number of valence electrons. This article will emphasize what a formal charge is, how to find the formal charge and its significance in practical applications. A formal charge does not represent an actual charge on an atom in a covalent bond but is used to predict the most likely structure when a compound has more than one valid Lewis’s structure. The aggregate formal charges on the atoms within a molecule or an ion must be equal to the overall charge on the molecule or ion. The charge distribution in a Lewis structure can be computed by formal charge formula. An atom in a molecule is given a formal charge (FC) based on the assumption that electrons in all chemical bonds are shared equally among atoms, regardless of relative electronegativity.įormal charges, also known as fake charges, are helpful for coordinate covalent bonding where one atom (the donor) gives both electrons to the acceptor atom. The formal charge usually “de-emphasize” the bond polarity by assuming that all electrons are shared equally. Moreover, it can explain the concept of resonance and draw Lewis structures representing resonance forms for a given molecule. 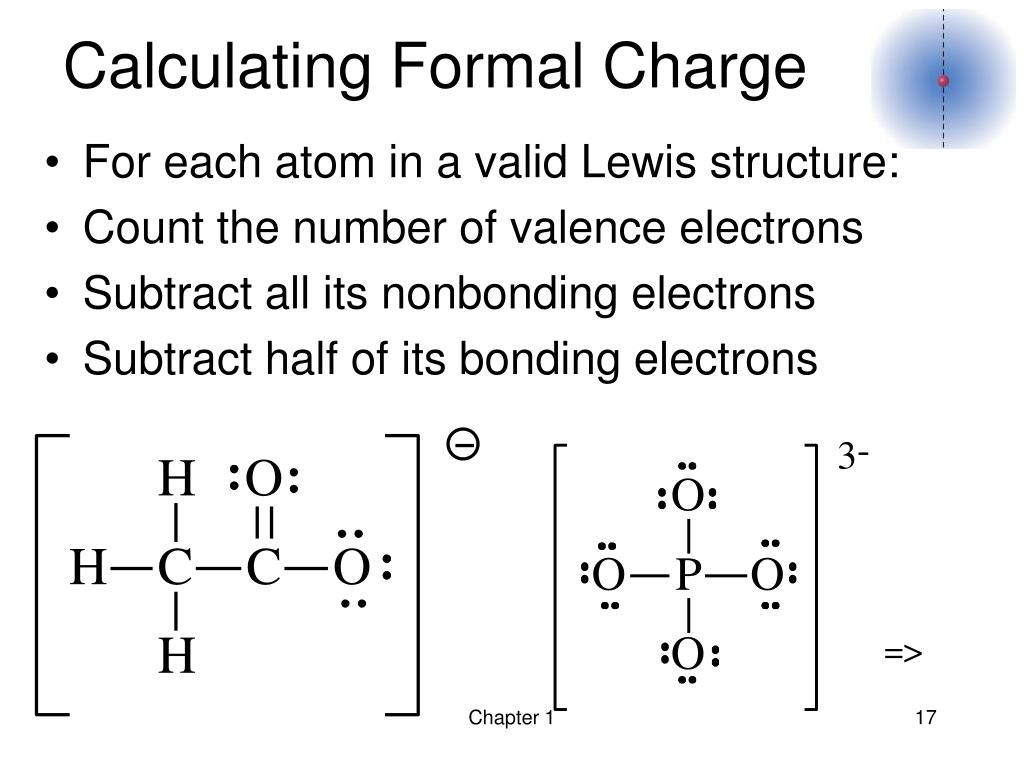
Learning the concept of calculating formal charges is essential because we can use formal charges to identify the most reasonable Lewis structures for a given molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
